Molecular diagnostic technology is a technology that uses DNA and RNA as diagnostic materials to detect the existence, defect, or abnormal expression of genes by using molecular biology techniques and then diagnosing human conditions and diseases. Its principle is to detect whether the structure of DNA or RNA changes, whether the amount and expression are abnormal, etc., to determine whether there are abnormal changes in the gene level of the subject. Molecular diagnostic technology is of great significance to preventing, predicting, diagnosing, and treating diseases. So what are the common techniques of molecular diagnosis?
1. What are the common techniques of molecular diagnosis?
2. The importance of IVD molecular diagnostic raw materials
3. Why is Yeasen capable of IVD RDC?
4. Customized product assortment
1. What are the common techniques of molecular diagnosis?
Molecular diagnostic techniques include quantitative PCR, nucleic acid sequence determination, molecular diagnostic techniques based on molecular conformation, and molecular diagnostic techniques of molecular hybridization.
1.1 Quantitative PCR (qPCR)
Compared with other molecular diagnostic detection technologies, nucleic acid amplification and detection of qPCR are carried out in the same closed system through fluorescent signals, to prevent the contamination of amplification products caused by uncapping after PCR. And by dynamically monitoring the fluorescent signal, low-copy templates can be quantified. Due to the above advantages, qPCR is currently the most accepted technology in clinical gene amplification laboratories. It is often used in the identification of pathogenic microorganisms such as viruses and bacteria, quantitative gene detection, gene polymorphism analysis, gene expression level monitoring, gene mutation screening, etc. However, the quality management issues related to this technology are becoming more and more prominent. The main problem facing qPCR today is how to eliminate the detection variation caused by various biological variables and reduce or suppress the interference factors in experimental operations and methodology. qPCR includes real-time fluorescence quantitative PCR, digital PCR, etc.
1.2 Molecular diagnostic technology of molecular hybridization
Southern blotting technology fragments DNA by restriction endonuclease and separates DNA fragments of different lengths by gel electrophoresis. Transfer to cellulose acetate membrane by siphon or voltage transfer, and then denature the DNA on the membrane and carry out molecular hybridization with nuclide-labeled oligonucleotide probes. After elution, autoradiography is used to identify the homologous sequence between the DNA fragment to be detected and the probe. This method has DNA fragment digestion and molecular probe hybridization and has high detection specificity, so it is the most classic molecular detection method in the field of probe hybridization. It can be widely used in the identification of various gene mutations, such as deletions, insertions, translocations, etc., and restriction fragment length polymorphisms.
The spot fixation technology of samples gets rid of the disadvantage of traditional Southern blotting requiring sample fixation by gel separation technology. By introducing a single base mutation into the plasmid vector, the first allele-specific oligonucleotide probe is constructed, making it possible to detect point mutations in nucleic acid sequences. Allele-specific oligonucleotide reverse dot blot(ASO-RDB) performs genotyping and mutation detection by hybridizing biotin-labeled specific PCR amplification products with probes immobilized on the membrane to develop color. This method can immobilize a variety of oligonucleotide probes on the same membrane strip and can screen hundreds of alleles of the DNA to be tested through only one hybridization reaction. The operation is simple and fast. ASO-RDB is a relatively common technology for gene mutation detection, genotyping, and pathogen screening.
1.3 Nucleic acid sequence determination
The only way to obtain nucleic acid sequence information is the sequencing reaction, which is also an important branch of molecular diagnostic technology. Currently includes 1st generation sequencing, 2nd generation sequencing, and 3rd generation sequencing. In 1975, Sanger and Coulson published a method for DNA sequence determination using the addition and subtraction method, which was the beginning of the era of nucleic acid sequencing. Sanger sequencing is the most classic first-generation sequencing technology, and it is also the most commonly used method to obtain nucleic acid sequences. Second-generation sequencing includes pyrosequencing and high-throughput second-generation sequencing. Pyrosequencing realizes the quantification of the base load ratio of specific sites and is widely used in SNP site detection, allele (mutation) frequency determination, and bacterial and virus typing detection. Currently, common high-throughput next-generation sequencing platforms mainly include Roche 454, Illumina, and ABI SOLiD. Both construct a DNA library by DNA fragmentation, amplify the library by cross-linking with the carrier, and carry out a reaction of synthesizing and sequencing on the carrier surface. The core concept of third-generation sequencing technology is to target single molecules by synthesizing and sequencing, which further reduces the cost and can perform single-molecule detection of mixed genetic materials.
1.4 Molecular diagnostic technology based on molecular conformation
Molecular diagnostic techniques based on molecular conformation include denaturing gradient gel electrophoresis, denaturing high-performance liquid chromatography, and high-frequency melting analysis.
2. The importance of IVD molecular diagnostic raw materials
Precision medicine is built based on molecular diagnostics. China's molecular diagnostic area is in a state of fast growth, thanks to governmental encouragement and technical iteration. How can businesses take advantage of this opportunity and improve their overall competitiveness?
From the perspective of downstream companies developing in vitro diagnostic reagents, The front line is extensive and winding from the standpoint of downstream firms manufacturing in vitro diagnostic reagents. It is time-consuming, labor-intensive, and costly, particularly during the product development stage. Traditional self-research sometimes requires a significant investment of time, money, and resources in terms of labor, equipment, and locations. At the same time, the product introduction is sluggish owing to unfamiliarity with the raw material characteristics and diagnostic reagent application method, making it difficult to assess product competitiveness.
To establish in-depth collaboration between upstream and downstream, mutual benefit, and win-win, downstream firms must pick suitable in vitro diagnostic raw material suppliers from the standpoint of upstream companies' in vitro diagnostic raw material suppliers.
2.1 Yeasen IVD RD——Customize IVD molecular diagnostic raw materials
Since 2014, Yeasen has supplied molecular enzymes for IVD diagnostic reagents. In addition to delivering high-quality and plentiful raw materials, it begins thinking based on the IVD industrial chain and intends to improve upstream and downstream collaboration. Rapid manufacturing helps IVD businesses to focus on core business.
Therefore, Yeasen created IVD RDC (IVD Research Development Center) to handle project demonstration, R&D design, product samples, small test and pilot test, and product finalization. Its core is to allow IVD enterprises to custom produce raw materials and debug products during R&D, finalize products quickly, and continue to deliver customized raw materials.
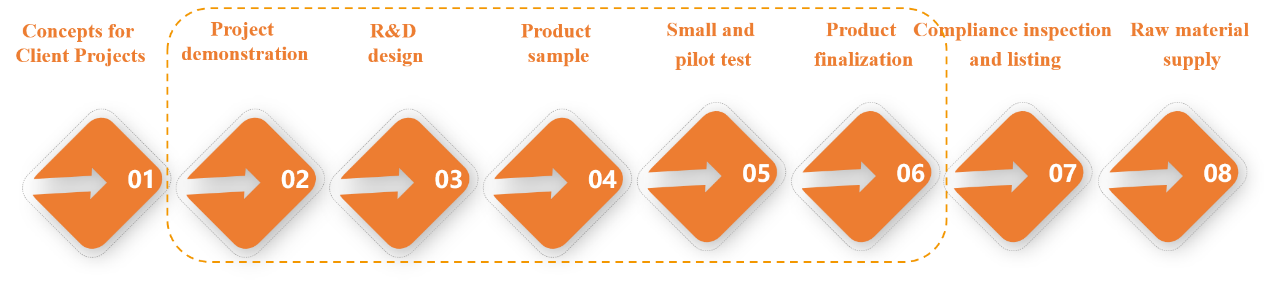
Figure 1. IVD RDC (IVD Research Development Center)
3. Why is Yeasen capable of IVD RDC?
Utilizing important technologies and R&D platforms, Yeasen has an effective R&D process and an exceptional team of professionals. The shortest time for product raw material creation and modification may be reduced from years, months, to weeks, allowing consumers to get their products sooner. The product is rapidly completed and released.
3.1 IVD RDC's primary platform

Figure 2. IVD RDC's primary platform
3.2 A critical manufacturing platform for IVD RDC: ISO-certified ultra-clean molecular enzyme production base—UCF.ME
Yeasen is equipped with industrial-grade AKTA purification and physical and chemical analysis equipment, a 100-level high-density fermentation unit, a 10,000-level standard clean workshop, and an automation production line, allowing for large-scale preparation.
- Produced in strict accordance with GMP standards;
- Acquired ISO 13485 accreditation for quality management systems;
- Have a separate manufacturing base: an ultra-pure molecular enzyme factory(UCF.ME®);

Figure 3. ISO 13485 certificate
3.3 Developing an effective R&D procedure for IVD RDC
IVD RDC does not operate in isolation; rather, it is backed by an entire industrial infrastructure. Yeasen Biotechnology Research Institute focuses on the evolution of enzymes and the discovery of new enzymes, establishing a link between production, education, and research, and establishing an enzyme library with the protein fermentation and purification platform of the Molecular Enzyme Research and Development Center to ensure a large number of optional raw materials based on market and customer demand. Orientation Enzymes are adjusted to continually increase the availability of raw materials. The high-throughput enzyme screening platform of the Molecular Enzyme R&D Center selects excellent proenzymes from a large number of candidate enzymes and delivers them to IVD RDCs.
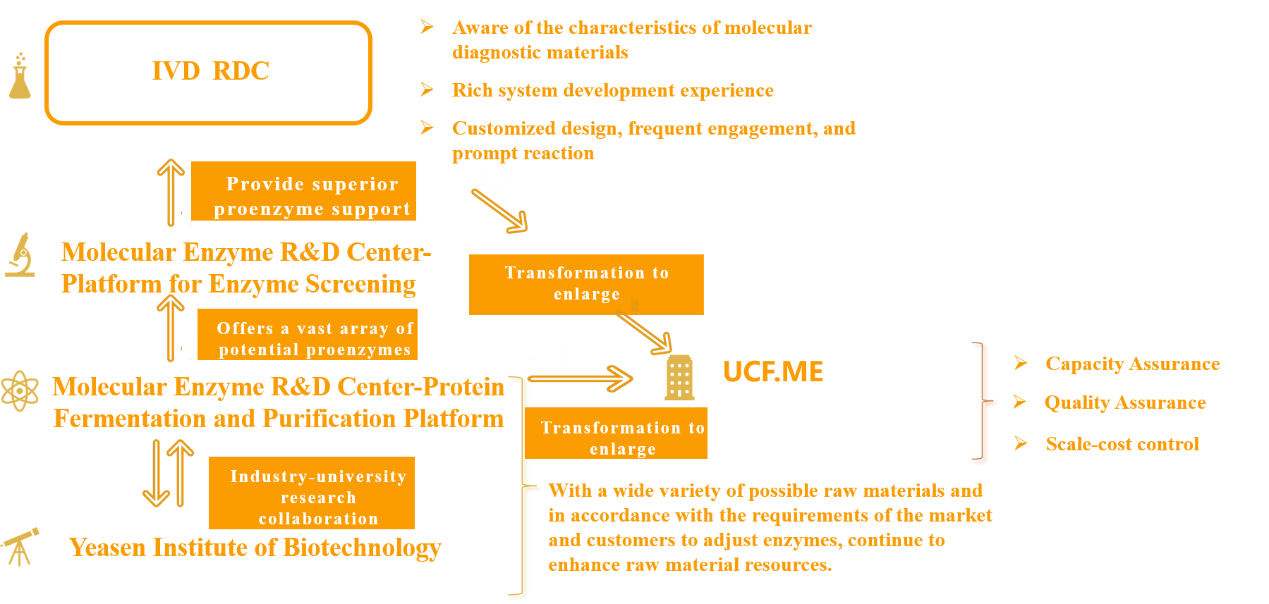
Figure 4. Efficient R&D Process
3.4 A competent group installing IVD RDC
The R&D personnel of Yeasen Biotechnology accounts for nearly 40%, Ph.D. and Master's make up roughly 90 percent of Yeasen Biotechnology's R&D workers, and they have exceptional development skills. IVD RDC is headed by project managers, which employs a matrix organizational structure, and organically merges R&D resources from diverse technological platforms to expedite development based on the specific demands of each client.
Customers gain from IVD RDC
Yeasen can offer customized solutions for individual product requirements and swiftly match the most appropriate raw materials, therefore optimizing product performance and enhancing product competitiveness; The UCF.ME of Yeasen will generate the product's raw materials to assure a continuous and steady supply of raw materials once the product's development has been completed, therefore reducing product development expenses.
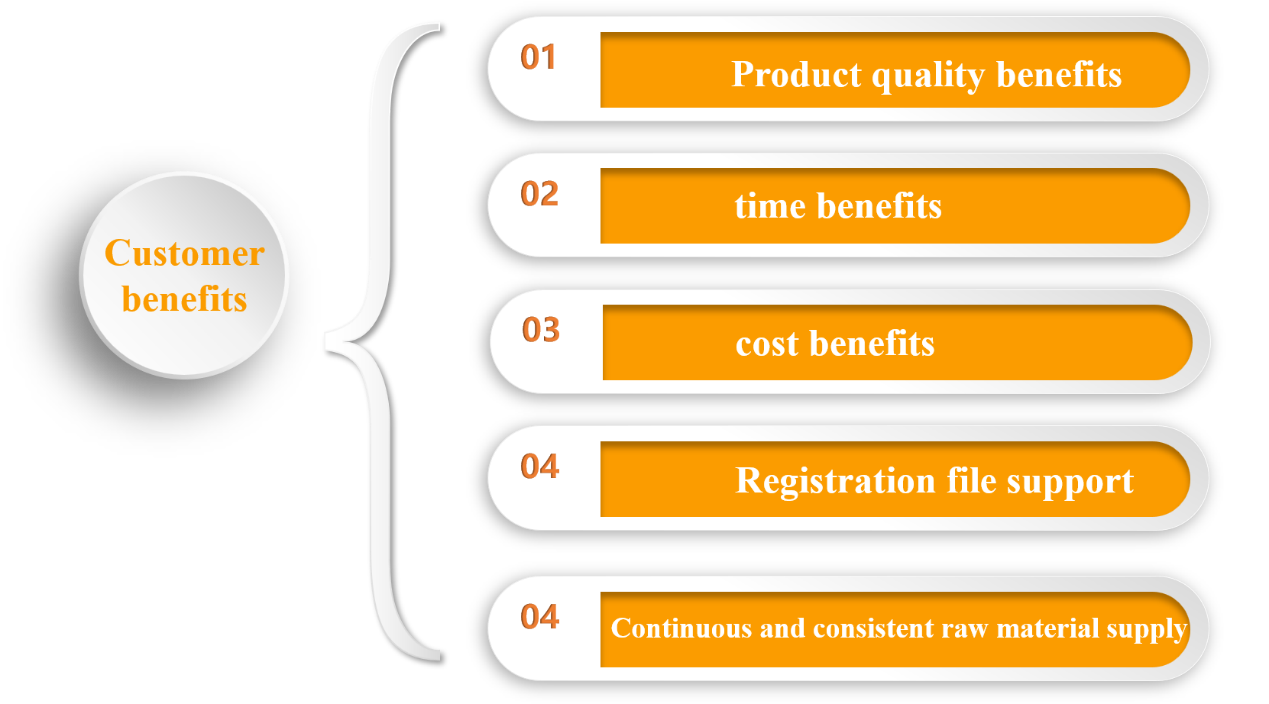
Figure 5. Customer benefits
4. Customized product assortment
4.1 Customizable product line
- Pretreatment of samples and nucleic acid extraction;
- PCR, RT-PCR, qPCR, RT-qPCR;
- Isothermal amplification;
- NGS;
- Freeze-dried raw materials and reagents;
- Others;
4.2 Customizable product categories
- Pretreatment of samples and nucleic acid extraction: Magnetic beads, single enzyme, buffer, etc.
- PCR, RT-PCR, qPCR, RT-qPCR: Single enzymes, buffers, master mixes, etc.
- Isothermal amplification: Single enzyme, buffer, chromogenic reagent, premix, etc.;
- NGS: Single enzyme, a module for assembling a library, etc.
- Freeze-dried raw materials and reagents: Lyophilization of a single enzyme, lyophilization of a component, lyophilization of a completed product, etc.;
- Others: Nucleic acid modification enzymes