The successful development of mRNA vaccines and drugs necessitates a diverse range of raw materials. As a leading innovator in the molecular enzyme industry, Yeasen has achieved success in producing various molecular enzymes through its advanced molecular enzyme directed evolution platform and state-of-the-art protein fermentation and purification technology platform.
In order to guarantee both product quality and production capacity, Yeasen has established a GMP-level production facility, catering to the complete industrial production requirements of its customers. With an annual production capacity exceeding 5 billion dosages, Yeasen is committed to meeting the demands of the market.
Moreover, Yeasen stands out as the pioneer in China to obtain the ISO 13485 certificate for the research, development, manufacture, and distribution of molecular enzymes. This certification ensures the quality, consistency, and traceability of all reagents, reinforcing Yeasen's dedication to maintaining the highest standards in the industry.
1. Advantage of GMP
2. Yeasen can provide GMP-grade Products
3. Yeasen Acquired DMF Numbers for Many Products
4. Application scenarios of Yeasen products
5. Raw materials for mRNA vaccine preparation provided by Yeasen
6. Ordering Information
1. Advantage of GMP
"GMP-grade" is a branding term that Yeasen uses to describe reagents manufactured at ISO 13485 certified facilities. Yeasen’s GMP-grade reagents are produced in compliance with ISO 13485 quality management system standards and with more stringent process controls and complete documentation records. Yeasen can not only offer high-quality reagents but also provide documents, site audits, and other support to our customers.
Table 1. Advantage of GMP
| Quality Controls | Research level | GMP-grade |
| Animal free | Animal-derived material may be used | Animal-free certificates can be provided if required |
| Cell bank characterization | Less stringent | Stringent (compliant with GMP regulations) |
| Traceability information for raw materials and finished product | Less stringent | Stringent (compliant with GMP regulations) |
| Endotoxin level control | / | Stringent |
| Sterile control | / | Stringent |
| Batch records for manufacturing and testing | Less stringent | Stringent (Batch records can beh provided if required) |
| Process validation report | / | The report can be provided if required |
| Change control system | Self-assessment | Stringent (compliant with GMP regulations) |
| Quality management system | Self-assessment | ISO 13485 certification |
| DMF number | / | Yes (reporting) |
2. Yeasen can provide GMP-grade Products
Based on wide experience and technical advantages in enzyme development and industrial production, Yeasen constructed a new facility, named "mRNAtools", to supply raw materials for mRNA-based drugs. Covering 50000 square feet, the mRNAtools facility is built and operated by GMP regulations and is equipped with 2×1500L automatic fermentation equipment, industrial-scale purification, and lyophilization equipment. To provide more strict reagents and also satisfy the production and registration requirements of customers. The molecular enzyme production base specially built for GMP-grade products ensures the quality of products from both hardware and software. 70 professional production team, team members have many years of GMP production experience. The warehousing and logistics are modularized. While ensuring the safety of product storage and transportation multi-center distribution across the country ensures the supply cycle.
3. Yeasen Acquired DMF Numbers for Core Products
Drug master files (DMFs) are documents submitted voluntarily to the US Food & Drug Administration (FDA) that contain confidential, detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of human drug products.
The FDA reviews the technical contents of a DMF when an API manufacturer incorporates it as a reference to support it's Investigational New Drug Application (IND), New Drug Application (NDA), Abbreviated New Drug Application (ANDA), and Export Application. DMFs have significant roles in the application. The FDA approval process is significantly shortened when using raw materials from manufacturers that have filed DMFs for these specific materials.
Yeasen has got the DMF files that can speed up the project schedule. You may request that we provide reference authorization to our DMFs in support of your application with the FDA. To initiate the reference authorization of our DMFs, please submit a Letter of Authorization request to Yeasen requesting that we provide a DMF Letter of Authorization to the respective FDA Center.
4. Application scenarios of Yeasen products
4.1 Template Generation
Plasmid linearization is an indispensable step during template generation when plasmid vectors are used as transcription templates. As transcription proceeds to the end of DNA templates, plasmid linearization ensures that RNA transcripts of a defined length and sequence are generated. Yeasen provides various restriction enzymes to meet your needs. We recommend selecting restriction enzymes that generate blunt ends or 5´-overhangs.
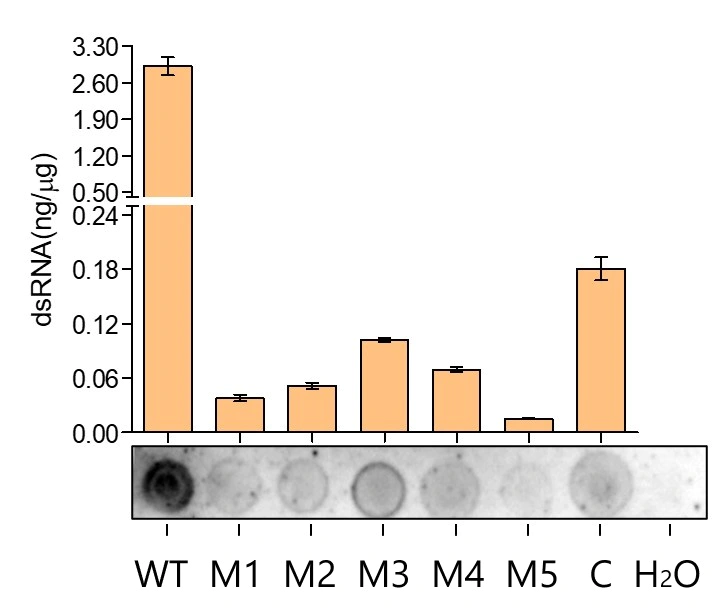
4.2 How to reduce dsRNA in Vitro Transcription ( IVT )?
In vitro transcription is affected by various factors. Enzymes, buffers, and modified substrates all have a great impact on transcription efficiency.
Based on the ZymeEditor platform, YEASEN Biotech has evolved novel T7 RNA polymerases which significantly reduce the generation of dsRNA during the in vitro transcription (IVT) process. It greatly lowers the immunogenicity of mRNA and, to a certain extent, enhances the integrity of mRNA. This development is expected to play a crucial role in advancing the development of mRNA vaccines and cancer drugs.
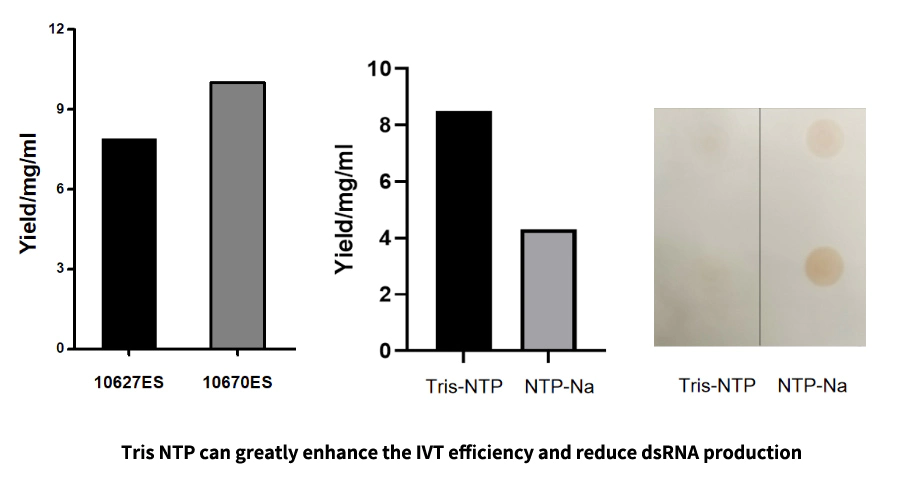
By optimizing the buffer and adjusting the salt type of the nucleotide substrate, we greatly improved the efficiency of transcription and reduced the production of dsRNA.
4.3 mRNA Capping
As a 5´ cap structure reduces immunogenicity and is required for efficient translation of mRNAs, adding cap structures to the 5′ ends of mRNA generated by transcription is necessary.
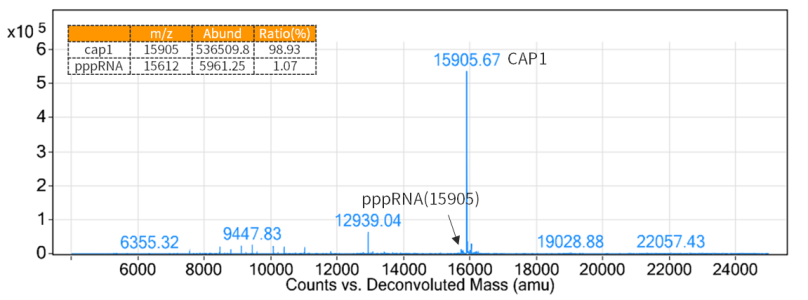
Figure 1. Cap rate detection results (LC-MS detection)
4.4 mRNA Purification
Purification is an indispensable step to achieving high-purity mRNAs.
5. Products for mRNA synthesis verified by Yeasen
At present, Yeasen can provide a complete set of products in the production process of mRNA stock solution. The products are strictly controlled by GMP production and fully meet the requirements of mRNA vaccine production.
Table 2. Raw materials for mRNA vaccine preparation provided by Yeasen
| Template preparation | In vitro transcription | Capping | Purification | ||||
| Hieff Canace™ Plus High-Fidelity DNA Polymerase (Inquiry) | 10153ES | T7 High Yield RNA Synthesis Kit | 10623ES | mRNA Vaccinia Capping Enzyme GMP-grade | 10614ES | Hieff NGS™ mRNA Isolation Master Kit | 12603ES |
| dNTP Set Solution | 10122ES |
10625ES 10628ES |
mRNA Cap 2´-O-Methyltransferase GMP-grade |
10612ES | Hieff NGS™ RNA Cleaner | 12602ES | |
| Hieff Clone™ Plus One Step Cloning Kit (Inquiry) | 10911ES | Pyrophosphatase,Inorganic GMP-grade | 10620ES | S-adenosylmethionine (SAM) GMP-grade | 10619ES |
metal magnetic stand (Inquiry) |
80461ES |
| Gold T4 DNA Ligase | 10300ES | Murine RNase inhibitor GMP-grade | 10621ES | Alkaline Phosphatase | Inquiry | ||
| Bsa I, BspQI GMP-grade, Xba I | 10661ES 10664ES 10662ES |
DNase I | 10611ES | CAP Analog | Inquiry | ||
| RNAse R (20U/uL) | 14606ES | NTP Set Solution | 10133ES | ||||
| MolPure™ Endo-free Plasmid Maxi Kit (Inquiry) | 19036ES | Pseudo UTP sodium solution GMP-grade (100 mM) | 10650ES | ||||
| N1-Me-Pseudo UTP sodium solution GMP-grade (100 mM) | 10651ES | ||||||
6. Ordering Information
The following are representative products offered by Yeasen. Additional sizes are available. Our products are highly optimized to work in concert, to help ensure superior performance and reproducibility. We can also provide customized services. If you’re interested in a product that isn’t shown, contact us and we’ll work with you to meet your needs.
Table 3. Ordering Information
Regarding reading:
GMP-grade reagents for mRNA in vitro synthesis
DNase I and Their Applications in Biomedicine
References:
[1] Chaudhary, N., Weissman, D., & Whitehead, K. A. (2021). mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nature reviews. Drug discovery, 20(11), 817–838.
[2] Vogel, A. B. et al. Self-Amplifying RNA Vaccines Give Equivalent Protection against Inflfluenza to mRNA Vaccines but
at Much Lower Doses. Molecular therapy : the journal of the American Society of Gene Therapy 26, 446-455,
[3] Fuchs, A. L., Neu, A. & Sprangers, R. A general method for rapid and cost-efficient large-scale production of 5'
capped RNA. RNA (New York, N.Y.) 22, 1454-1466, doi:10.1261/rna.056614.116 (2016).
[4] Schmid, A. Considerations for Producing mRNA Vaccines for Clinical Trials. Methods in molecular biology (Clifton, N.J.) 1499, 237-251,
[5] Banerji, A. et al. mRNA Vaccines to Prevent COVID-19 Disease and Reported Allergic Reactions: Current Evidence and Suggested Approach. The journal of allergy and clinical immunology. In practice 9, 1423-1437,
[6] Richner, J. M. et al. Modifified mRNA Vaccines Protect against Zika Virus Infection. Cell 168, 1114-1125.e1110,
[7] Corbett, K. S. et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 586,567-571,
[8] Corbett, K. S. et al. mRNA-1273 protects against SARS-CoV-2 beta infection in nonhuman primates. Nature immunology 22, 1306-1315,
[9] Keech, C. et al. Phase 1-2 Trial of a SARS-CoV-2 Recombinant Spike Protein Nanoparticle Vaccine. The New England journal of medicine 383, 2320-2332,
[10] Kramps, T. & Elbers, K. Introduction to RNA Vaccines. Methods in molecular biology (Clifton, N.J.) 1499, 1-11,
[11] Linares-Fernández, S., Lacroix, C., Exposito, J. Y. & Verrier, B. Tailoring mRNA Vaccine to Balance Innate/- Adaptive Immune Response. Trends in molecular medicine 26, 311-323,