SuperSignal SuperDura Extended Duration Substrate
Description
YEASEN ECL series luminescence Kit is designed to detect antibodies and associated antigens directly or indirectly labeled with horseradish peroxidase (HRP). The principle is that proteins or nucleic acids were transferred to the imprinted membrane after electrophoresis, and the target proteins on the membrane were bound by primary antibody and secondary antibody labeled with HRP, or the nucleic acids on the membrane were bound directly or indirectly by probes labeled with HRP. After washing the membrane, the ECL working solution prepared by the product was used to incubate the membrane at room temperature for several minutes. The imprinted membrane was wrapped with plastic wrap and fixed to the X-ray exposure Cassette. Then the X-ray film is pressed on the membrane in a darkroom and exposed for several seconds to several hours. After development and fixing, protein or nucleic acid bands can be clearly displayed on the X-ray film.
This kit has a unique luminescent/enhanced substrate system, and Western blotting detection of medium fick-grade can be achieved. In addition to X-ray, can also be detected by fluorescent CCD.
Features
- High sensitivity and high signal-to-noise ratio, it can detect medium fick-grade antigens.
- The signal lasts for 24 hours, and the luminescence time is more than 10 times longer than that of conventional ECL substrate. High-quality imprinted images can be obtained by multiple exposures for publication.
- Higher dilution ratio of antibodies can be used to greatly save antibodies.
- Dilution ratio of primary antibody (liquid storage concentration 1mg/ mL) : 1:1000-1:50000.
- Dilution ratio of secondary antibody (liquid storage concentration 1mg/ mL): 1:50000-1:250000.
Applications
- Chemiluminescence ELISA
- Western Blot
- Dot Blot-DNA/RNA
- Southern blot-DNA
- Northern blot-RNA
Specifications
| Recommended Antibody Concentrations | primary antibody (liquid storage concentration 1mg/mL): 1:1000-1:50000 secondary antibody (liquid storage concentration 1mg/mL): 1:50000-1:250000 |
| Signal Duration | < 24 h |
| Substrate Properties | Chemical Substrate |
| Substrate Type | HRP (Horseradish Peroxidase) Substrate |
| Sensitivity | medium fick-grade antigens |
Components
| Components No. | Name | 36223ES10(10 mL) | 36223ES60(100 mL) | 36223ES70(200 mL) | 36223ES76(500 mL) |
| 36223-A | SuperDura ECL -Reagent A | 5 mL | 50 mL | 100 mL | 250 mL |
| 36223-B | SuperDura ECL -Reagent B | 5 mL | 50 mL | 100 mL | 250 mL |
Storage
The product should be stored at 2℃~8℃ for one year.
Notes: 36223-A should be stored away from the light!
Figures
- Effect Contrast of Extended Duration Substrate
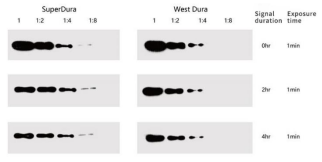
Figure 1. Comparison of detection effect between YEASEN SuperDura substrate and West dura of the same level substrate of other brands.
YEASEN ECL reagent has a better use effect than other brands of the same level of products
[1] Tan Q, Lin S, Zeng Y, et al. Ginsenoside Rg3 attenuates the osimertinib resistance by reducing the stemness of non-small cell lung cancer cells. Environ Toxicol. 2020;35(6):643-651. doi:10.1002/tox.22899(IF:3.118)
Catalog No.:*
Name*
phone Number:*
Lot:*
Email*
Country:*
Company/Institute:*

